Bcs Classification Of Drug
Contents. BCS classes According to the Biopharmaceutical Classification System (BCS) drug substances are classified to four classes upon their solubility and permeability:. Class I - high, high. Example:. Those compounds are well absorbed and their absorption rate is usually higher than excretion.
Jump to CASE STUDIES I-II—APPLICATION OF BCS IN INNOVATOR DRUG. - Pregabalin is a BCS Class 1. Respect to drug and excipients. The Biopharmaceutics Classification System (BCS) is a scientific framework for classifying drug substances based on their aqueous solubility and intestinal permeability.
Class II - high permeability, low solubility. Example:, paracetamol,.
The of those products is limited by their solvation rate. A correlation between the bioavailability and the solvation can be found. Class III - low permeability, high solubility. Example:. The absorption is limited by the permeation rate but the drug is solvated very fast.
If the formulation does not change the permeability or gastro-intestinal duration time, then class I criteria can be applied. Class IV - low permeability, low solubility. Example:. Those compounds have a poor bioavailability. Usually they are not well absorbed over the intestinal mucosa and a high variability is expected.
Definitions The drugs are classified in BCS on the basis of solubility, permeability, and dissolution. Solubility class boundaries are based on the highest dose strength of an immediate release product. A drug is considered highly soluble when the highest dose strength is soluble in 250 ml or less of aqueous media over the pH range of 1 to 7.5. The volume estimate of 250 ml is derived from typical bioequivalence study protocols that prescribe administration of a drug product to fasting human volunteers with a glass of water.
Permeability class boundaries are based indirectly on the extent of absorption of a drug substance in humans and directly on the measurement of rates of mass transfer across human intestinal membrane. Alternatively non-human systems capable of predicting drug absorption in humans can be used (such as in-vitro culture methods). A drug substance is considered highly permeable when the extent of absorption in humans is determined to be 90% or more of the administered dose based on a mass-balance determination or in comparison to an intravenous dose.
For dissolution class boundaries, an immediate release product is considered rapidly dissolving when no less than 85% of the labeled amount of the drug substance dissolves within 15 minutes using USP Dissolution Apparatus 1 at 100 RPM or Apparatus 2 at 50 RPM in a volume of 900 ml or less in the following media: 0.1 N HCl or simulated gastric fluid or pH 4.5 buffer and pH 6.8 buffer or simulated intestinal fluid. See also. References.
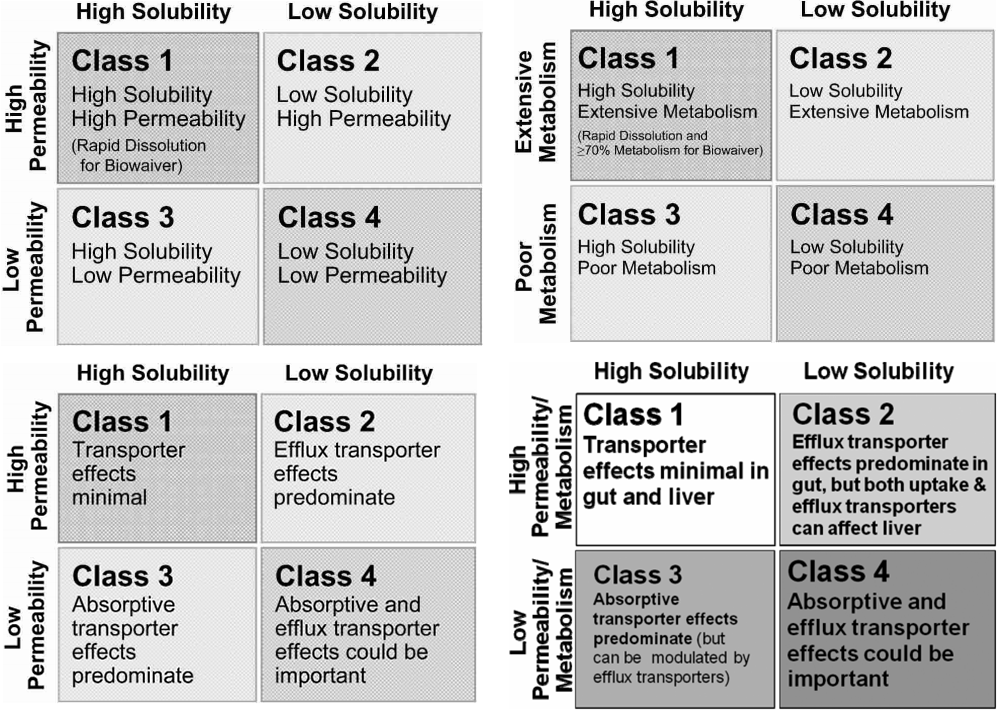
Biopharmaceutics Classification System and Biopharmaceutics Drug Distribution Classification System are complimentary, not competing, classification systems that aim to improve, simplify, and speed drug development. Although both systems are based on classifying drugs and new molecular entities into four categories using the same solubility criteria, they differ in the criterion for permeability and have different purposes. Here, the details and applications of both systems are reviewed with particular emphasis of their role in drug development. © 2012 Wiley Periodicals, Inc. And the American Pharmacists Association J Pharm Sci 102:34–42, 2013.
INTRODUCTION The United States Food and Drug Administration's (US FDA) Biopharmaceutics Classification System (BCS) is based on the work of Amidon et al. With the core idea being that in vitro methodology, centrally embracing permeability, and solubility, with qualifications related to pH and dissolution, may qualify drug products for a waiver of in vivo bioequivalence studies. The objective of the BCS is to predict in vivo performance of drug products from in vitro measurements of permeability and solubility. In 2005, Wu and Benet recognized that for drugs exhibiting high intestinal permeability rates, the major route of elimination in humans was via metabolism, whereas drugs exhibiting poor intestinal permeability rates were primarily eliminated in humans as unchanged drug in the urine and bile. They proposed that a Biopharmaceutics Drug Disposition Classification System (BDDCS) could serve as a basis for predicting the importance of transporters in determining drug disposition, as well as in predicting drug–drug interactions. The major differences between BCS and BDDCS relate to their purpose and the measurement for classification as depicted in Table. The purpose of BCS is to characterize drugs for which products of those drugs may be eligible for a biowaiver of in vivo bioequivalence studies.

The purpose of BDDCS is to predict drug disposition and potential drug–drug interactions in the intestine and the liver, and potentially the kidney and brain. Both BCS and BDDCS use solubility as one of the two classification criteria. The solubility parameter utilized may be called the US FDA solubility, that is, an estimate of the ability of the drug at its highest dose strength to completely dissolve in 250 mL of water over a pH range between 1 and 7.5 at 37°C. For a drug to be considered highly soluble in the two classification systems, the drug from its highest strength regulatory approved dosage form must go completely into solution at its lowest solubility over this pH range in 250 mL of water.
As we have recently noted, US FDA solubility is a property of the drug in a formulation and is not an intrinsic property of the active pharmaceutical ingredient itself. The second classification parameter, and where the two systems differ, is related to intestinal permeability. In BDDCS, predictions are based on intestinal permeability rate, which was found to be related to extent of drug metabolism. In BCS, biowaivers are based on the extent of intestinal absorption, which in a number of cases does not correlate with intestinal permeability rate. THE BCS AND ITS USE IN DRUG DEVELOPMENT The BCS characterizes drugs into four classes according to their US FDA solubility and permeability as depicted in Figure. In 2000, the US FDA promulgated the BCS system as a science‐based approach to allow waiver of in vivo bioavailability and bioequivalence testing of immediate‐release solid oral dosage forms for Class 1 high solubility, high‐permeability drugs when such drug products also exhibited rapid dissolution. This waiver is based on a triple‐tier rationale where: (a) high solubility insures that drug solubility will not limit dissolution, and thus absorption, (b) high permeability insures that drug is completely absorbed during the limited transit time through the small intestine, and (c) rapid dissolution insures that the gastric emptying process is the rate‐limiting step for absorption of highly soluble and highly permeable drugs.
Drug sponsors are allowed to use mass balance, absolute bioavailability, or human intestinal perfusion studies to demonstrate high permeability. The US FDA Guidance, however, also recommends possible methods not involving human subjects including in vivo or in situ intestinal perfusion in a suitable animal model, and/or in vitro permeability methods using excised intestinal tissues or monolayers of suitable epithelial cells, usually the Caco‐2 cell system. However, some studies have shown that in vitro cellular permeability criteria recognized in the US FDA's BCS guidance may not always correctly predict the extent of drug absorption in humans. The Biopharmaceutics Classification System (BCS) as defined by the US FDA after Amidon et al.
In 2010, the European Medicines Agency (EMA) revised its bioequivalence guideline stating that demonstration of complete absorption in humans is preferred for biowaiver of BCS Class 1 drug applications rather than measures of high permeability. The criterion for complete absorption in the EMA Guideline is ≥85% measured extent of absorption in humans based either on absolute bioavailability or mass balance studies. The correlation between intestinal permeability rate and the extent of absorption in humans came from the results of in vivo studies with 34 drugs and endogenous substances, where a good correlation was observed between jejunal permeability from human intestinal perfusion studies and the fraction of the oral dose absorbed in humans. However, in these early human intestinal perfusion studies, no drugs were investigated that subsequently showed a discordance between cellular system permeability rates and the extent of absorption in humans.

It is now generally recognized by the US FDA, the EMA, and the research scientists in the field that high cellular permeability rates do correctly predict a high extent of absorption. Discordance is essentially only found for some nonmetabolized drugs exhibiting low cellular permeability rates but complete absorption. The role of BCS in drug development is facilitating the possibility of obtaining a waiver of in vivo bioequivalence studies for drug products, where the regulatory agencies recognize the drug as BCS Class 1 and where the dissolution rate of the new drug product meets the rapid dissolution criteria of the regulatory agencies. This is definitely a simplifying and cost saving procedure in drug development.
However, there is no predictive benefit to BCS. Studies in humans must be carried out to show that the drug achieves complete absorption (≥90% for US FDA and ≥85% for EMA, although the US FDA has indicated informally that ≥85% may be appropriate for a biowaiver). Yet, as stated above, cellular studies exhibiting high‐permeability rates can give sponsors confidence that a high‐solubility compound will meet the extent of absorption criteria of the regulatory agencies before obtaining actual extent of absorption measures in humans.
Since the promulgation of the US FDA BCS Guidance in 2000, a number of new possible class boundaries have been proposed for additional biowaivers. For example, the EMA will grant biowaivers for BCS Class 3 drugs with high solubility but limited absorption. In addition, a World Health Organization (WHO) Technical Report suggests that biowaivers may be appropriate for so‐called BCS Class 2a drugs, weak acids that exhibit low solubility only at low pH.
The WHO recommended criteria for such drug products would be rapid dissolution at pH 6.8 and a similar dissolution profile to the innovator product at pH 1.2, 4.5, and 6.8. Like the EMA, the WHO Technical Report also recommends biowaiver eligibility for Class 3 very rapidly dissolving drug products that contain no inactive ingredients that are known to alter GI motility and/or absorption.
However, at this time only BCS Class 1 drugs are eligible for a biowaiver of in vivo bioequivalence from the US FDA and BCS Class 1 as well as some Class 3 drugs by the EMA. BDDCS As described above, the purpose of BDDCS is to predict drug disposition and potential drug–drug interactions in the intestine and the liver with an emphasis on defining which drugs would be amenable to enzymatic‐only and transporter‐only disposition and drug–drug interactions, as well as where transporter–enzyme interplay may be important.
Recent reviews from the Benet Lab - have defined these enzymatic, transporter, and transporter‐interplay characteristics with potential transporter effects following oral dosing as depicted in Figure. The recognition of the correlation between BCS intestinal permeability and BDDCS extent of metabolism by Wu and Benet preceded an explanation for these findings.
We hypothesize now that high‐permeability‐rate compounds are readily reabsorbed from the kidney lumen and from the bile, facilitating multiple accesses to the metabolic enzymes. For example, consider the BCS/BDDCS Class 1 drug letrozole. This completely oral available drug is primarily eliminated by metabolism via CYP3A4 and CYP2A6 enzymatic processes with less than 4% of the dose excreted unchanged in the urine. However, letrozole is only 60% bound to plasma proteins and thus it might be expected, based on glomerular filtration rate and fraction unbound, that renal clearance could approach 48 mL/mL. Yet the total clearance for letrozole is only 40.5 mL/min with less than 4% excreted unchanged. Thus, this high‐permeability compound is reabsorbed in the kidney tubules (and possibly from the bile) with the major route of elimination being metabolic processes.
The rationale for the correlation between intestinal permeability rate and the extent of metabolism appears to be based on the fact that high‐permeability‐rate compounds are reabsorbed from potential unchanged drug excretion routes in the body and thus can only be eliminated through metabolism. This hypothesis then led us to conclude that the measure of high‐permeability rate in making the BDDCS assignment need not necessarily be a human biological membrane or membrane surrogate, but that passive permeability in any appropriate membrane model may provide the correct assignment. This topic will be discussed further below. Predict potential drug–drug interactions not tested in the drug approval process. Predict the potential relevance of transporter–enzyme interplay. Assist the prediction of when and when not transporter and/or enzyme pharmacogenetic variants may be clinically relevant.
Predict when transporter inhibition by uremic toxins may change hepatic elimination. Predict the brain disposition. Increase the eligibility of drugs for BCS Class 1 biowaivers using measures of metabolism. For drugs already in the market, BDDCS provides potential predictability of drug–drug interactions that had not been anticipated or tested in the drug approval process. For example, our laboratory recognized that atorvastatin was a BDDCS Class 2 drug exhibiting extensive metabolism and poor solubility.
Thus, as shown in Figure, we recognized that atorvastatin may potentially exhibit a drug–drug interaction with inhibitors of hepatic uptake transporters. We first demonstrated in cellular and isolated perfused rat liver studies that atorvastatin, as well as its two active hydroxylated metabolites, were substrates for human and rat organic anion transporting polypeptides (OATPs) and that inhibition of OATP uptake would decrease atorvastatin metabolism. We then carried out whole animal studies in rats to confirm this finding in intact animals.
We then demonstrated in healthy volunteers that a single intravenous (i.v.) dose of rifampin, a potent OATP inhibitor would significantly increase the total area under the curve (AUC) of atorvastatin acid by 6.8 ± 2.4‐fold and that of 2‐hydroxy‐atorvastatin acid and 4‐hydroxy‐atorvastatin acid by 6.8 ± 2.5‐fold and 3.9 ± 2.4‐fold, respectively. Of course, once recognizing that atorvastatin and its active metabolites are substrates for OATP1B1, then it would logically follow that genetic variants in this transporter would affect atorvastatin pharmacokinetics, as has been demonstrated. Thus, BDDCS is also useful in predicting where pharmacogenetic variants can yield meaningful drug disposition changes.
In a further study, we demonstrated that inhibiting the hepatic uptake transporter for glyburide would also significantly increase its AUC and that blood glucose levels were lower than those observed after dosing with glyburide alone. Glyburide is primarily a substrate of OATP1B3, which does not exhibit significant changes in activity with genetic variants, and therefore, one would suspect that a pharmacogenetic study of the transporter would not yield significant changes. However, glyburide is a substrate for CYP2C9, with known disposition changes for the genetic variants of this enzyme. It is important to recognize that the BDDCS characterization of transporter effects and transporter enzyme interplay as depicted in Figure does not predict that every drug in each class will display the effects listed. Rather, BDDCS predicts what transporter effects may occur, and which may not, and what should be tested. As an example, the Class 2 drug felodipine is a CYP3A substrate, but not a substrate for P‐glycoprotein (P‐gp), and we used it as our control in examining Class 2 drug efflux transporter–enzyme interplay. Furthermore, one cannot be sure that a cellular transporter–enzyme interaction will translate into an in vivo clinically relevant interaction, even when the in vitro K i values suggest that the interaction needs to be tested.
As an example, numerous publications concerning the pharmacogenomics of warfarin have shown that accounting for the genetic variants of CYP2C9 and VKORC1 plus other patient parameters can only explain about 55% of the variability observed for this drug in patient populations. Because warfarin is a Class 2 drug, we asked could it be a substrate for an uptake transporter, and if so might knowledge of this transporter genotype increase the predictability? Rat and human hepatocyte studies showed that warfarin appeared to be a substrate for OATP uptake that could be inhibited by rifampin, which might account for an approximately 30% change in AUC. The in vitro interaction was of the same magnitude as what we had observed in vitro for glyburide. We then carried out a human study that showed that there was no significant increase in warfarin blood concentrations in the presence of the OATP inhibitor rifampin. Recently, the US FDA has recommended that studies in renal failure patients be carried out even for drugs where renal elimination of unchanged drug is minimal. This recommendation comes about in part based on a finding that was related to the development and characterization of BDDCS.
Previous studies of changes in drug metabolism in renal failure patients for drugs primarily eliminated by hepatic metabolism were thought to be related to the effects of uremic toxins as either potential inhibitors or down regulators of metabolic enzymes. However, this could be tested in vitro and was shown not to occur in many cases. We began to recognize that previously unexplained effects of renal disease on hepatic metabolism can result from accumulation of substances (toxins) in renal failure that modify hepatic uptake and efflux transporters, - and that this mechanism could explain why BDDCS Class 2 drugs could demonstrate changes in metabolism in renal failure, whereas this would not be observed for BDDCS Class 1 drugs when in vitro uremic toxins did not alter microsomal metabolism.
To demonstrate the relevance of the potential effect of uremic toxins in patients, we compared the pharmacokinetics of oral and i.v. Erythromycin in patients with end stage renal disease versus healthy volunteers. Erythromycin is a BDDCS Class 3 drug that is primarily eliminated unchanged in the bile. It is a substrate for hepatic uptake transporters that we had previously shown can be inhibited by uremic toxins.
We demonstrated that the hepatic clearance of erythromycin in end stage renal disease patients was decreased by 31% ( p = 0.01) and that bioavailability was increased 36%. Because we had given the drug both i.v. And orally, we calculated that there was no change in the fraction of the oral dose absorbed multiplied by the potential gut availability ( F abs F g). This would be expected even though erythromycin, a Class 3 BDDCS drug is a substrate for an intestinal uptake transporter, as it is not possible for the uremic toxins to be present in the intestine. Thus, the BDDCS allows investigators to predict the potential effect of uremia on hepatic metabolism and biliary excretion. To facilitate use of the BDDCS system for making predictions for drugs on the market, we recently compiled the BDDCS classification for 927 drugs, which include 30 active metabolites.
Of the 897 parent drugs, 707 (78.8%) drugs are administered orally. Where the lowest measured solubility was found in the literature, this value was reported for 72.7% (513) of these orally administered drugs.
Measured values are reported for the percentage excreted unchanged in the urine, log P and log D 7.4, when available. For all 927 compounds, the in silico parameters for predicted log solubility in water, calculated log P, polar surface area, and the number of hydrogen bond acceptors and hydrogen bond donors for the active moiety are also provided, thereby allowing comparison analyses for both in silico and experimentally measured values.
Fda Guidance Bcs Classification
We showed that when comparing the in silico parameters across the four classes, there is a distinct difference between Class 2 and Class 3 compounds. However, surprisingly the log P and solubility in silico parameters for the Class 1 drugs appear to be intermediate between those for Class 2 and Class 3 and not very different than the parameters for the Class 4 drugs. We note this failure of in silico parameters to efficiently predict whether a drug will be Class 1, believed by many to be the most desirable because of high solubility and high permeability, versus Class 4 drugs that are low solubility and low permeability.
Most recently, we have shown that the prediction of brain disposition of orally administered drugs may be improved using BDDCS. It is generally believed that high log P, high permeability, and lack of P‐gp efflux are desirable characteristics for central nervous system (CNS) drug candidates to become marketed CNS drugs. From the literature, we were able to identify 153 marketed drugs that met three criteria: (a) central or lack of central pharmacodynamics effects were known, (b) the BDDCS class was identified, and (c) information was available as to whether the drug was or was not a substrate for P‐gp. About 98% of BDDCS Class 1 drugs were found to be markedly distributed throughout the brain; this includes 17 BDDCS Class 1 drugs known to be P‐gp substrates. Thus, we expand upon the transporter effects listed in Figure for Class 1 drugs. We now believe that transporter effects are minimal, and clinically insignificant, for Class 1 drugs in the gut, liver, and brain, and suspect that this is also true for the kidney.
General Classification Of Drugs
Recognition that BDDCS Class 1 drugs that are P‐gp substrates will still yield central effects allowed us to decrease the number of compounds incorrectly predicted from in silico parameters in terms of disposition from the 19%–23% employing previous methods, to less than 10%. This finding of a lack of a clinically significant effect of P‐gp on brain disposition of BDDCS Class 1 drugs has marked implications for predicting drug disposition and effects of new molecular entities (NMEs), as will be described in the last section of this report. Finally, there is also an application to BCS biowaivers inherent in the BDDCS Classification System.
Recognized that for 29 drugs where measured human intestinal permeabilities were available, the extent of metabolism correctly predicted high versus low permeability for 27 of 29 (or 93%) of the measured human intestinal permeabilities in terms of BCS. Thus, Benet and coworkers recommended that regulatory agencies add the extent of drug metabolism (i.e., ≥90% metabolized for US FDA and ≥85% metabolized for EMA) as an alternate method for determining the extent of drug absorption in defining Class 1 drugs suitable for a waiver of in vivo studies of bioequivalence. The authors propose that the following criteria be used to define the ≥90% metabolized for US FDA marketed drugs: “Following a single oral dose to humans, administered at the highest dose strength, mass balance of Phase I oxidative and Phase II conjugative drug metabolites in the urine and feces measured as either unlabeled, radioactive‐labeled or nonradioactive‐labeled substances, account for ≥90% of the drug dose.
This is the strictest definition for a waiver based on metabolism. For an orally administered drug to be ≥90% metabolized by Phase I oxidative and Phase II conjugative process, it is obvious that the drug must be absorbed.” In their 2010 revised bioequivalence guidance, EMA incorporated this recommendation.
US FDA scientists have also supported this recommendation, although no formal written guidance change has been issued. Predict the major route of elimination of an NME in humans (metabolism vs. Excretion of unchanged drug in the urine and bile). Predict the relevance of transporters and transporter–enzyme interplay in drug disposition as detailed in Figure 2 and Table 2. Predict central or lack of central effects. Predict the effects of high‐fat meals on the extent of bioavailability.
For an NME, it would be most useful to predict its BDDCS class before any studies in humans, animals, or even cellular systems. The recognition of the correlation between intestinal permeability rate and extent of metabolism allows prediction of BDDCS class for an NME to be based on passive membrane permeability. Initially, we proposed to follow the BCS permeability rate measure in the Caco‐2 cellular system using metoprolol, but now more preferably labetalol, as the cutoff between high and low permeability compounds. However, because we now believe that it is the passive permeability that is the predictive parameter, we suggest that even studies with an artificial membrane such as parallel artificial membrane permeability assay (PAMPA) will provide a reasonable prediction of BDDCS Class 1 and Class 2 versus BDDCS Class 3 and Class 4 using labetalol as the cutoff marker. We evaluated the permeability results for 21 drugs studied by three different PAMPA models (a lipid/oil/lipid trilayer, a biomimetic, and a hydrophilic filter membrane PAMPA assay). For these 21 drugs, the human extent of absorption and metabolism was known.
In this evaluation, the extent of absorption or metabolism was defined as being low or high if it was less than 30% or ≥90%, respectively. Permeability was defined as being low or high if it was less than 2.0 or ≥3.5 × 10 −6 cm/s, respectively, based on a marked differentiation using these cutoffs for 19 of the 21 drugs.
The high PAMPA permeability for all three models accurately predicted BCS ≥90% absorption very well and only slightly less accurately BDDCS metabolism. However, for low PAMPA permeability, although the system very accurately predicted poor BDDCS metabolism, the systems only correctly predicted absorption 25% of the time. On the basis of these data, we suggest that passive transcellular drug permeability in an artificial membrane may reasonably predict extensive versus poor human metabolism.
Note that we are not suggesting that the permeability rate cutoffs listed above are numbers that may be translated to other laboratories. They are just the values from our in vitro permeability rate analyses used to attempt to differentiate the 21 drugs for which human in vivo permeability rate measures and extent of metabolism have been reported. Although we believe it is quite easy, and using high‐throughput methods, to predict with good accuracy Class 1 and Class 2 versus Class 3 and Class 4 assignment of an NME, it is not easy to differentiate high solubility from low solubility.
The major impediment is that the BDDCS (and BCS) solubility criterion is based on the highest marketed dose strength. Of course, for an NME such knowledge may be years away from the compounds initial evaluation. Therefore, we propose following the recommendation of Pfizer scientists to use a solubility cutoff of 200 μg/mL (i.e., 50 mg highest dose strength) in the initial evaluation. Thus, compounds with a lowest solubility over the pH range 1–7.5 being greater than 200 μg/mL would be assigned Class 1 or Class 3 and those with a lowest solubility less than 200 μg/mL would be assigned Classes 2 and 4. As we have noted previously, in silico predictions of solubility are not very reliable and thus we also recommend following the Pfizer protocol of utilizing a high‐throughput equilibrium solubility assay for NMEs in simulated gastric fluid at pH 1.2 and in 50 mM phosphate buffer pH 6.5, for the initial assignment.
Thus, very early in the drug development of an NME, using only in vitro methods to assign BDDCS class, sponsors will be able to predict the major route of elimination of an NME in humans (metabolism versus excretion of unchanged drug in the urine and bile) and predict the relevance of transporters and transporter–enzyme interplay in drug disposition. If an NME is BDDCS Classes 1 or 2, the drug should exhibit good absorption, but not necessarily good bioavailability. In contrast, if the NME is BDDCS Classes 3 or 4, then good absorption will only be achieved if the NME is a substrate for an intestinal uptake transporter or possibly small enough to pass through intestinal pores.
There is a marked difference in the BDDCS class distribution of drugs on the market as opposed to NMEs. We previously estimated the distribution for NMEs based on all recently synthesized medicinal compounds. In Figure, we depict the distribution of oral immediate‐release drugs on the market versus small molecule NMEs, the latter percentages determined from a data set of Professor Oprea encompassing 28,912 medicinal chemistry compounds tested for at least one target and having affinities of micromolar or less concentrations. Although 40% of oral immediate‐release marketed drugs are Class 1, only 18% of NMEs fall in this category. This difference is primarily related to Class 2 drugs, where 33% of marketed oral immediate‐release products are found versus 54% of NMEs. As can be seen in Figure, quite similar numbers between marketed drugs and NMEs are found for Classes 3 and 4.
That is, in essence, NMEs are becoming larger, more lipophilic and less soluble, with time in the drug discovery paradigm. Distribution of 698 oral immediate‐release drugs on the market and NME percentages from a data set of 28,912 medicinal chemistry compounds tested for at least one target and having affinities at micromolar or less concentrations using BDDCS criteria. Early characterization of the BDDCS class of an NME offers information related to all those characteristics listed in Table for drugs on the market. Of particular relevance, early in drug development, may be the information related to potential brain distribution as described in our recent publication. That is, if central effects for an NME are not desired, then a BDDCS Class 2 compound that is a substrate for brain efflux transporters would be preferable to a highly soluble BDDCS Class 1 NME. On the contrary, sponsors should recognize that if the NME was, in fact, BDDCS Class 1, then brain distribution and potential central off‐target effects cannot be avoided even if the compound is shown to be a substrate for brain efflux transporters.
BDDCS classification may also allow predictions regarding food effects for orally dosed drugs. The AUC and bioavailability of many drugs are greatly affected by concomitant food intake and the US FDA recommends that high‐fat meals (800–1000 cal; 50%–65% from fat, 25%–30% from carbohydrates, 15%–20% from protein) may be used in food effect studies in humans.
Many factors are believed to contribute to these food effects, including changes in gastric emptying time, bile flow, pH of the intestine, splanchnic blood flow, and gut wall metabolism. A variety of evidence exists supporting food effects on transporters as well, as described by Custodio et al. In general, high‐fat meals have no effect on the extent of absorption of BDDCS Class 1 drugs, increase A.
Comments are closed.